TB vaccine may reduce risk of atopy in infants
- John Evans
- Aug 27, 2021
- 1 min read
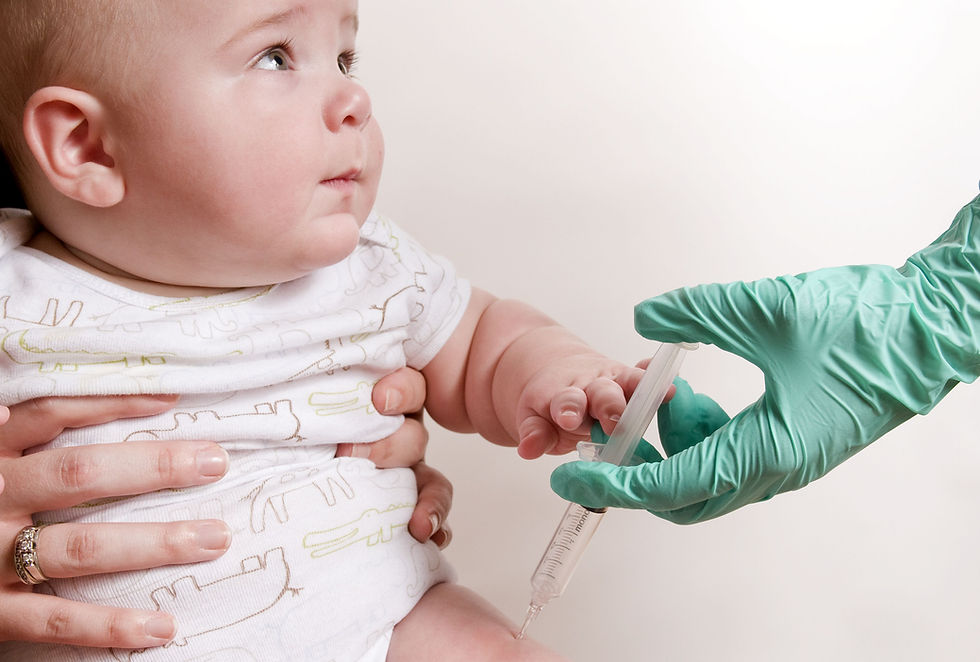
A neonatal dose of the Bacille Calmette-Guérin (BCG) tuberculosis vaccine could reduce the risk of atopic disease in infants with two atopic parents, according to findings from an Australian study.
The rationale for the study was the idea that BCG vaccination may stimulate the immune system, making it better able to combat infections and less prone to allergies, the study's authors note.
A press release from the Murdoch Children's Research Institute in Victoria, Australia notes that the vaccine is roughly a century old and had been used to prevent tuberculosis in Australia until the late 1980s.
To test the potential for BCG vaccination to be protective against atopic disease, researchers conducted a randomized controlled trial with 1,272 infants. The participating infants were randomized to receive either the BCG vaccine or no BCG vaccine at birth. The investigators then evaluated 12-month incidence of eczema based on questionnaires conducted every three months, as well as with a clinical visit at 12 months.
The initial findings were published online ahead of print in Allergy (July 26, 2021). In those findings, the BCG vaccine had a modest beneficial effect in preventing eczema in high-risk infants.
The benefit was greater in infants with two atopic parents—those who have hay fever, asthma or eczema. In these infants, a single dose of BCG soon after birth reduced the incidence of eczema by 25%.
However, the authors noted there is insufficient evidence to recommend neonatal BCG vaccination for all infants to prevent eczema in the first year of life.
This cohort will be followed through their first five years of life, according to the release.



Comments