Study of sebaceous gland stem cells reveals insights into abnormal cell growth
- By John Evans
- Aug 2, 2019
- 2 min read
Photo by: OpenStax College
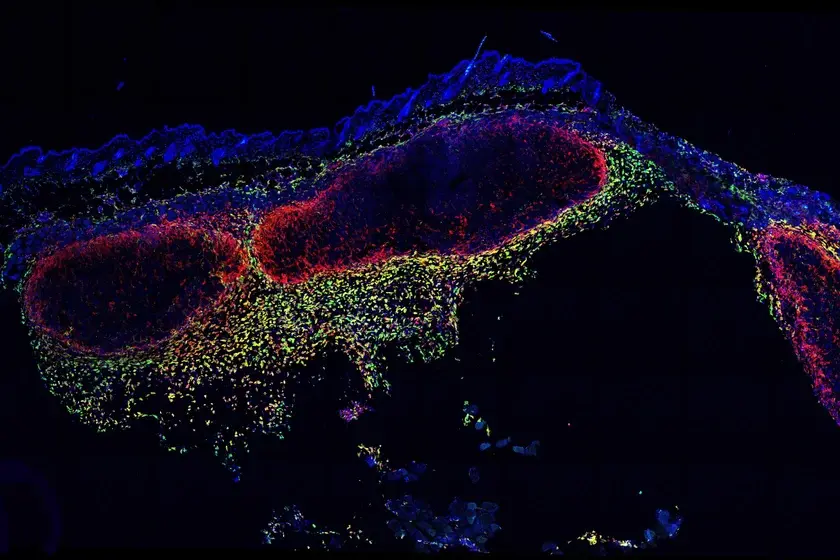
A study of stem cells in the development and life of sebaceous glands has shed new light into how these structures form in the skin, how they are replenished during their operational life, and how certain cancer-inducing mutations impact stem cells in these glands. In the paper, published in Nature Cell Biology (July 29, 2019; 21:924–932), the researchers applied large-scale quantitative fate mapping—tracing the life of cells from their position in embryonic tissue to their location in adult tissue—in mouse models to define the patterns of cell fate behaviour during the development and maintenance of stem cells. “We demonstrate for the first time ever how the sebaceous glands that contribute to the natural moisture of the skin are formed and how they are maintained throughout life by stem cells,” said lead author Marianne Stemann Andersen in a press release. “This knowledge may be transferred to individuals with sebaceous gland conditions, e.g. acne or very dry skin.” Andersen is a postdoctoral student in the Biotech Research & Innovation Centre (BRIC) at the University of Copenhagen. The authors also demonstrated how the behaviour of the stem cells changes when they introduced a specific and frequently found cancer mutation—the oncogene KrasG12D—to the skin stem cells. Unexpectedly, the mutation did not cause the cells to divide more often. Instead, the stem cells had a tendency to generate more stem cells rather than mature sebaceous gland cells when they divided. Healthy stem cells in immature sebaceous glands would more frequently divide into two more stem cells, until the gland was fully formed. After that, the stem cells would typically divide to replace mature sebaceous gland cells that had burst to release lipids. With the mutation, the stem cells tended to maintain their immature state of producing more stem cells than mature sebocyte cells. “In this case, the result is a sebaceous gland which—similar to tumours—continues to grow,” said senior author Kim B. Jensen, PhD. “We hope this knowledge can contribute to the design of better cancer treatment,” said Dr. Jensen, an associate professor from BRIC and the Novo Nordisk Foundation Center for Stem Cell Biology (DanStem) at the University of Copenhagen. This finding has implications for cancer treatments, many of which target rapidly-dividing cells. This research shows that abnormal stem cell differentiation, even at normal rates of division, can result in abnormal tissue growth, the authors note.




Comments