Melanoma research: GPER may explain why previous pregnancies could be advantage against melanoma
- by Bianca Quijano
- Jan 24, 2018
- 2 min read
The G protein-coupled estrogen receptor (GPER), once activated and combined with anti programmed death(PD)-1 inhibitor therapies, extended survival rate and eliminated tumours in 50% of cancer mouse models that have had three consecutive pregnancies, according to a study published in the journal eLIFE (Jan. 16, 2018).
This trial showed that much of the melanoma protective effect likely results from estrogen signalling of GPER on melanocytes, which is activated during pregnancy.
The investigators noted that this may explain why female sex and pregnancy hormones provide an advantage against melanoma. It may also introduce a new approach to cancer therapy.
“So much of the cancer field is focused on inhibitors, but in this new treatment approach, we’re actually activating something rather than blocking it,” said Dr. Todd W. Ridky, senior study author and assistant professor of dermatology at the University of Pennsylvania in Philadelphia, in a press release. “We used a synthetic compound to mimic part of what happens naturally during pregnancy, and as a result, the GPER activator is very well tolerated without any obvious toxic side effects that are common with most cancer drugs.”
The scientists engineered primary human melanocytes with lentiviruses to express mutant oncoproteins, commonly associated with spontaneous human melanoma including BRAFV600E (doxycycline-inducible), dominant-negative p53R248W, active CDK4R24C and hTERT. Then the oncogenic cells were combined with primary human keratinocytes and native human dermis. Out of this combination, they created three-dimensional human skin tissues that were grafted on the backs of female mice.
The mice were separated into two experimental arms: non-breeding or breeding groups.
After 15 weeks human tissues were harvested and analyzed. Grafts from the non-breeding group developed into melanocytic neoplasms. In contrast, tissues from the breeding group contained primarily non-proliferating melanocytes that were confined to the basal epidermal layer.

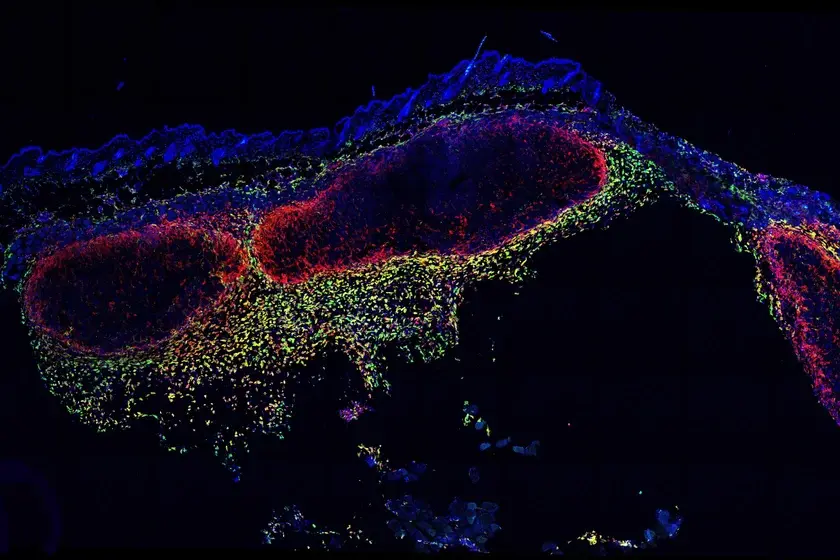
This diagram illustrates the methods utilized and results seen in the study. Figure B compares melanoma progression in the non-breeding and breeding mice. It shows the histologic characterization of representative orthotopic skin and resulting tumours, including hematoxylin and eosin (H/E), melanocyte and proliferation markers MITF, Ki67/MART, and Fontana Masson (melanin) among the non-breeding mice. Photo by Natale et al., courtesy of eLIFE.
The therapy induced long-term changes characterized by increased expression of well-established melanocyte differentiation antigens, increased pigment production, decreased proliferative capacity, and decreased expression of the oncodriver and stem cell marker c-Myc.
These long-lasting phenotypic changes inhibited tumour growth and also rendered tumour cells more susceptible to clearance by native immune cells. This ultimately made melanoma cells more vulnerable to immunotherapy.
“We hope this work inspires other researchers to revisit old ideas of differentiation-based cancer therapies now that immune therapies are available,” said Dr. Ridky. “It is clear that the future of cancer therapy lies in combination treatments, and differentiation drivers may be a very useful component in future cancer therapy regimens.”
Dr. Ridky said this method could be effective for other types of cancers.
“This is a receptor that is expressed in many organs, so there’s a reasonable expectation that this may work in other tumour types too,” said Dr. Ridky.
His team is planning further toxicity studies before advancing into human trials.


Comments